|
7/16/2023 0 Comments Moles to particles converter
The simplest type of manipulation using molar mass as a conversion factor is a mole-mass conversion (or its reverse, a mass-mole conversion). 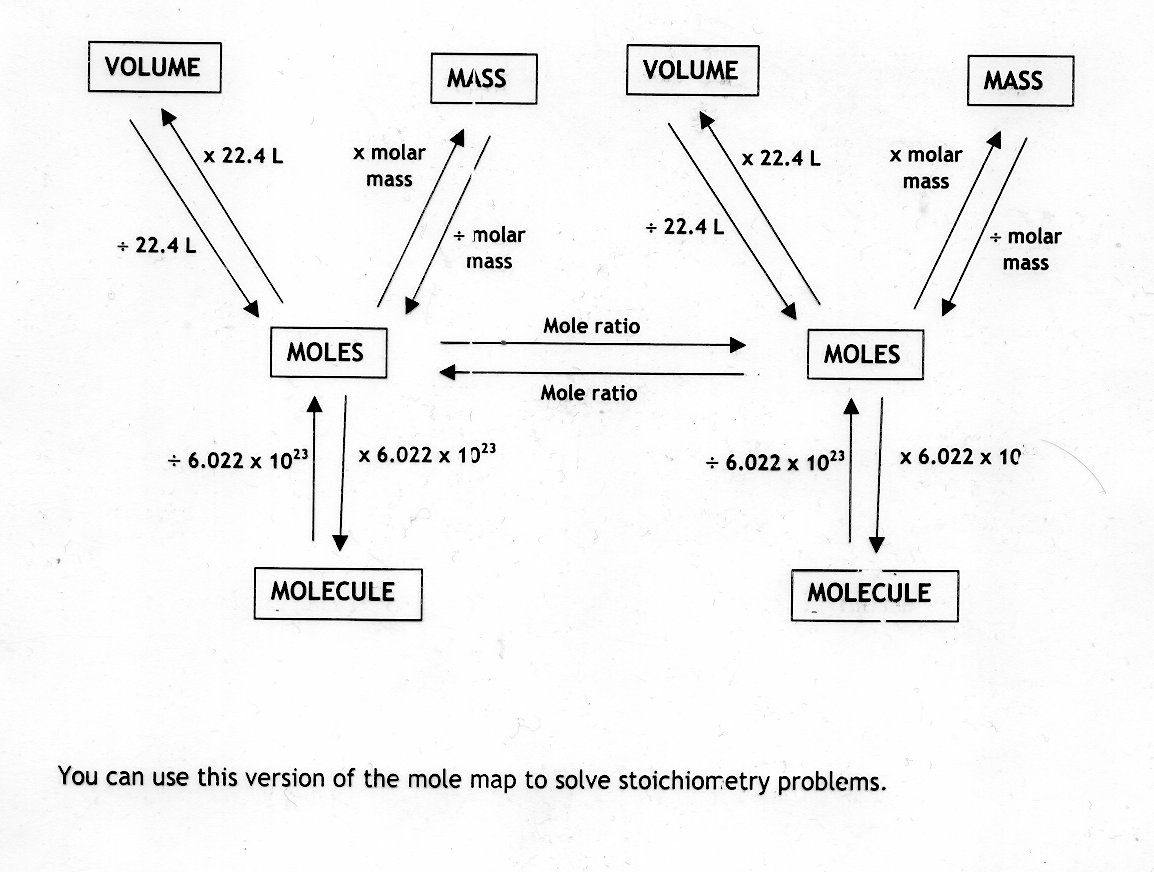
This relationship is frequently used in the laboratory. Now do the conversion directly:Īuthor: Fred Senese Chemistry Online! How can I convert moles to milliliters?Ĭomments & questions to Revised 02/23/18. The representative particles can be atoms, molecules, or formula units of ionic compounds. To find the number of milliliters that 1.00 mol of helium would occupy at 298 K and 1.00 atm, compute the molar volume But if you don't know the density, and the gas can be treated as ideal, you can calculate the molar volume of the gas from the ideal gas law and use it to convert moles to liters directly. If you know the density of the gas, you can use the first conversion method For example, to find the number of milliliters of 0.123 M AgNO 3 solution that contains 1.00 mol of AgNO 3,Ĭonvert moles of AgNO 3 to liters of solution using the molarity:Ĭonverting moles of gas to milliliters of gas. You'll need to know the concentration of the solute in the solution to do this conversion. Here's the basic strategy:įor example, to find the number of milliliters that 10.0 moles of NaCl would occupy, you must find the molecular weight (58.45 g/mol) and look up the density (2.17 g/mL):Ĭonverting moles of solute to milliliters of solution. You must know the substance's atomic or molecular weight and its density to do this conversion. Before you can make the conversion, ask: moles of what ? Milliliters of what?Ĭonverting moles of a substance to milliliters of the same substance. Algebra Civil Computing Converter Demography Education Finance Food Geometry Health Medical Science Sports Statistics. 
There are at least three different mole-to-volume conversions you'll encounter in general chemistry. Use this simple science mole to molecules calculator to calculate molecule.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
